Introduction:
Involvement of the central nervous system (CNS) remains a challenge in childhood B-cell precursor acute lymphoblastic leukemia (BCP-ALL). Currently, the investigation of CNS leukemia mechanisms relies heavily on 2D cell culture and mouse models. However, given the differences between human and murine CNS in cellular identity and architecture, it becomes crucial to explore alternative models to study CNS leukemia. Moreover, novel targets for diagnosis and treatment of CNS ALL are critically needed.
Methods:
In this study, we established a pioneering 3D co-culture model that combines human induced pluripotent stem cell (iPSC)-derived cerebral organoids with BCP-ALL cells. We developed new methods to extract data from our invasion assays by enhancing the visualization of 3D organoid images, enabling us to accurately measure the invasion depth of leukemia cells compared to healthy controls within the organoids relative to their surface. To gain further insights on leukemia cells invading the organoids compared to those in the non-invaded fraction, we conducted RNA sequencing and immunofluorescence staining. Subsequently, we validated these results in a BCP-ALL in vivo mouse model and in patients initially diagnosed with CNS-positive BCP-ALL compared to CNS-negative cases within a cohort of 100 BCP-ALL patients.
Results:
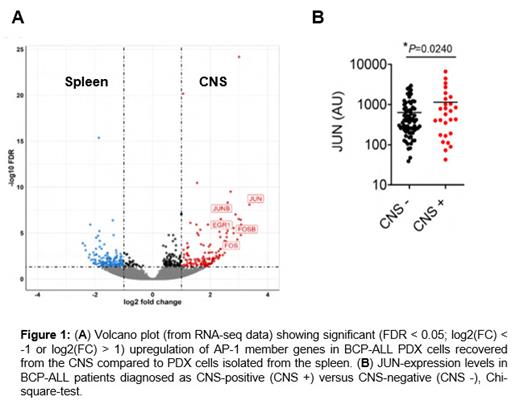
Our experiments demonstrated robust and deep engraftment of TCF3::PBX1+ leukemia cell lines and patient-derived xenograft (PDX) cells into cerebral organoids (within a 14-day of co-culture). In contrast, the engraftment of healthy human CD34+ hematopoietic stem and progenitor cells (HSPCs) was limited ( p < 0.05) as compared to leukemia cells. Utilizing the co-culture model, we successfully validated the targeting of CNS leukemia-relevant pathways, such as CD79a/Igα or CXCR4-SDF1, via genetic knockdown or blocking experiments using inhibitor, respectively, which reduced the invasion of BCP-ALL cells into the organoids. Of note, RNA sequencing and immunofluorescence staining analysis revealed a significant upregulation of members of the AP-1 transcription factor complex, namely FOS, FOSB, and JUN, in the organoid-invading cells. Furthermore, we found an enrichment of AP-1 pathway genes in PDX cells recovered from the CNS compared to spleen blasts of mice transplanted with TCF3::PBX1+ PDX BCP-ALL cells (n = 5), thereby supporting the critical role of AP-1 signaling in CNS disease (Figure 1A). In line with these findings, we observed significantly higher levels of the AP-1 gene JUN in patients initially diagnosed with CNS-positive BCP-ALL compared to CNS-negative cases (mean JUN expression: 633.4 ± 78.51 in CNS-negative vs 1142 ± 296.5 in CNS-positive) within a cohort of 100 BCP-ALL patients (Figure 1B).
Summary:
In summary, we present ALL co-culture systems with iPSC-derived cerebral organoids as a promising complementary model to investigate CNS involvement in BCP-ALL including therapeutic targeting approaches, and identified the AP-1 pathway as a marker of CNS disease in TCF3::PBX1+ BCP-ALL.
Disclosures
Cario:Servier, Amgen: Research Funding; JazzPharma: Speakers Bureau.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal